Chemia supramolekularna, czyli chemia „ponad cząsteczką”, to interdyscyplinarna dziedzina badań na pograniczu chemii, biologii i fizyki. Chemia supramolekularna zajmuje się badaniem zjawisk, w których wiodącą rolę odgrywają oddziaływania międzycząsteczkowe, takich jak rozpoznanie molekularne i samoorganizacja, stanowi więc fundament biologii z jednej, a nanotechnologii z drugiej strony.
Naszą specjalnością jest synteza organiczna, którą aplikujemy w dwóch głównych obszarach: w chemii supramolekularnej anionów oraz do konstrukcji tzw. Metal Organic Frameworks (MOFs), czyli krystalicznych polimerów koordynacyjnych posiadających w strukturze krystalicznej puste przestrzenie o rozmiarach nanoskopowych (luki, kanały, etc.).
Obszary zainteresowania
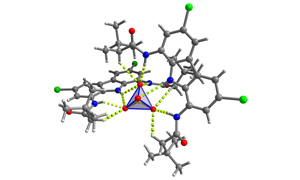
Aniony odgrywają fundamentalną rolę w chemii, biologii, medycynie i ochronie środowiska. W naszym laboratorium zajmujemy się projektowaniem, syntezą i badaniem właściwości kompleksotwórczych molekularnych receptorów na aniony.
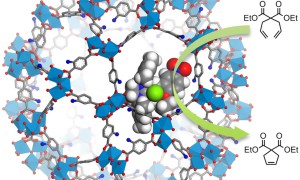
MOF-y, czyli Metal-Organic Frameworks, to regularne, porowate i łatwe do modyfikacji struktury, które mogą stanowić unikalne środowisko dla katalizy; środowisko, które można porównać do miejsc aktywnych enzymów…